Composition of ciprofloxacin drops
Cipro eye and ear drops contain ciprofloxacin in the form of ciprofloxacin hydrochloride. Each milliliter of solution contains 3.5 mg ciprofloxacin hydrochloride, which corresponds to 3 mg of pure ciprofloxacin.
In addition to the active substance, the formulation contains several excipients with specific technological and safety roles. These include benzalkonium chloride as a preservative, acidity regulators such as sodium acetate, acetic acid, sodium hydroxide, or concentrated hydrochloric acid to maintain the desired pH, stabilizers such as mannitol and disodium edetate, and purified water as the solvent.

The combination of active ingredient and excipients is designed to ensure chemical stability of ciprofloxacin, comfort during instillation into the eye or ear canal, and reliable antimicrobial preservation of the multidose bottle. Patients with known hypersensitivity to benzalkonium chloride or other components should consult a healthcare professional before use.
This medication is primarily available as eye and ear drops marketed under brand names such as Ciloxan. These topical preparations are formulated specifically for ophthalmologic (eye) and otologic (ear) use.
Oral tablets containing ciprofloxacin, including products sold under the names Baycip and Cipro, represent different dosage forms with distinct indications, systemic exposure, and dosing regimens. They should not be confused with or substituted directly for ophthalmic or otic drops without medical supervision.
Mechanism of action
Ciprofloxacin belongs to the class of antibiotics known as quinolones, more specifically the fluoroquinolones. Its primary antibacterial effect is bactericidal, meaning it kills susceptible bacteria rather than merely inhibiting their growth.
The core mechanism of action involves interference with bacterial DNA synthesis. Ciprofloxacin selectively inhibits the bacterial enzyme DNA gyrase (also referred to as topoisomerase II), which is essential for supercoiling and replication of bacterial DNA. By blocking this enzyme, ciprofloxacin prevents cell division and leads to rapid bacterial cell death.
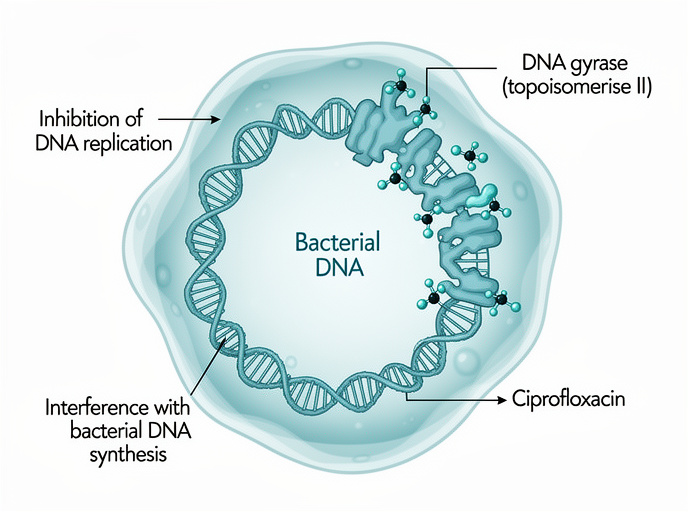
This targeted mechanism explains the high activity of ciprofloxacin against a wide range of Gram-negative and Gram-positive organisms and its usefulness in treating infections of the cornea, conjunctiva, outer ear canal, and middle ear with tympanostomy tubes.
Antibacterial spectrum
Ciloxan (ciprofloxacin drops) is highly active against most Gram-negative bacteria, including Pseudomonas aeruginosa, and many aerobic Gram-positive bacteria such as staphylococci and streptococci. Its in vitro activity has been extensively documented for both ocular and otologic pathogens.
For ocular infections, confirmed susceptible Gram-positive aerobes include Staphylococcus aureus (including methicillin-resistant strains), other staphylococci such as S. epidermidis, various corynebacteria, Streptococcus pneumoniae (pneumococcus), and viridans group streptococci. Confirmed Gram-negative aerobes include Acinetobacter, Haemophilus influenzae, Pseudomonas aeruginosa, and Moraxella species.
Additional ocular pathogens considered susceptible based on systemic inhibition zone diameter (IDD) criteria and minimum inhibitory concentrations (MICs) of ≤ 1 μg/mL in vitro include:
- Aerobic Gram-negative microorganisms: Acinetobacter calcoaceticus, Enterobacter aerogenes, Escherichia coli, Haemophilus parainfluenzae, Klebsiella pneumoniae, Neisseria gonorrhoeae, Proteus mirabilis, Proteus vulgaris, and Serratia marcescens.
- Other susceptible organisms: Peptococcus spp., Peptostreptococcus spp., Propionibacterium acnes, and Clostridium perfringens.
Non-susceptible organisms include some strains of Burkholderia cepacia, Stenotrophomonas maltophilia, and certain anaerobic bacteria, particularly Bacteroides fragilis, which may show resistance to ciprofloxacin.
Ophthalmic indications and efficacy
Ciprofloxacin eye drops are indicated for corneal ulcers and superficial infections of the eye and its adnexa caused by ciprofloxacin-susceptible bacteria. Clinical trials in patients with acute bacterial eye infections have confirmed high efficacy against a broad range of pathogenic microorganisms.
Ciprofloxacin has demonstrated in vitro activity against many additional ocular pathogens; however, the clinical relevance of these findings is not always fully established. Specifically, for some microorganisms, the safety and efficacy of ciprofloxacin in treating corneal ulcers or conjunctivitis have not been confirmed in adequately controlled clinical studies.
The relationship between systemic in vitro susceptibility parameters such as IDD or MIC and clinical outcomes after topical ophthalmic administration has not been fully defined. Nonetheless, ciprofloxacin concentrations measured in tear film, corneal tissue, and the anterior chamber after topical use are often ten to several hundred times higher than the MIC90 values for sensitive ocular pathogens, supporting its clinical effectiveness.
Otologic indications and efficacy
In otology, ciprofloxacin ear drops are used for the treatment of acute otitis externa (inflammation and infection of the external ear canal) and acute otitis media with tympanostomy tube drainage when caused by susceptible organisms. Clinical trials in patients with acute otitis externa have confirmed high activity against a wide spectrum of bacteria responsible for ear infections.
Topical ciprofloxacin has been shown to be active in vitro against most strains of the following microorganisms implicated in ear infections, although for some species the full clinical significance remains to be clarified:
- Aerobic Gram-positive microorganisms: Bacillus spp., Corynebacterium spp., Enterococcus faecalis, Staphylococcus aureus, Staphylococcus haemolyticus, Streptococcus pneumoniae, and viridans group streptococci.
- Aerobic Gram-negative microorganisms: Achromobacter xylosoxidans subsp. xylosoxidans, Acinetobacter baumannii, Acinetobacter junii, Acinetobacter lwoffi, Acinetobacter radioresistans, Acinetobacter genospecies 3, Citrobacter freundii, Citrobacter koseri, Enterobacter aerogenes, Enterobacter cloacae, Escherichia coli, Haemophilus influenzae, Klebsiella oxytoca, Klebsiella pneumoniae, Moraxella catarrhalis, Proteus mirabilis, Pseudomonas stutzeri, and Serratia marcescens.
For otitis media, ciprofloxacin has also shown in vitro activity against Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pneumoniae, Escherichia coli, Haemophilus influenzae, Moraxella catarrhalis, and Pseudomonas aeruginosa. As with ocular use, the correlation between systemic susceptibility criteria and clinical outcomes after topical otic administration is not fully established.
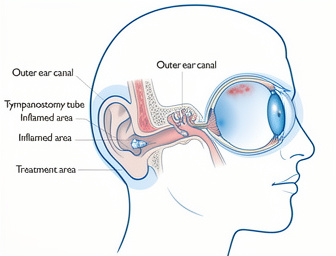
Overall, ciprofloxacin ear drops provide a convenient option for localized therapy in ear infections, achieving high concentrations at the site of infection with minimal systemic exposure. Frequent medical follow-up is recommended to ensure optimal response and to allow timely modification of treatment if necessary.
Resistance and cross-resistance
Resistance to ciprofloxacin generally develops slowly; however, parallel resistance within the class of gyrase inhibitors has been observed. Susceptibility studies show that many ciprofloxacin-resistant bacteria are also resistant to other fluoroquinolones.
In clinical trials of ciprofloxacin eye and ear drops, the incidence of isolates that acquired resistance during therapy was low. Due to the unique mode of action, there is typically no cross-resistance between ciprofloxacin and antibacterial agents with different chemical structures, including beta-lactam antibiotics, aminoglycosides, tetracyclines, macrolides, peptides, sulfonamides, trimethoprim derivatives, and nitrofurans. Consequently, bacteria resistant to these drug classes may still be susceptible to ciprofloxacin.
Preclinical safety data
Like other quinolones, ciprofloxacin has been shown to cause arthropathy in young animals of most species studied after oral administration. At a dose of 30 mg/kg, joint changes were minimal. This exposure is approximately 270 times higher than the recommended clinical ear dose for treating a 10 kg child with 0.27 mg ciprofloxacin in each ear twice daily.
In a one-month study in young beagle dogs, no joint damage was observed after topical application of Ciloxan eye drops, and there was no evidence that local ocular therapy adversely affected joints. Clinical and radiographic evaluation of 634 children who received oral ciprofloxacin likewise showed no skeletal toxicity.
Reproduction studies in rats and mice using doses 50 times the maximum daily ophthalmic dose for humans and 900 times the recommended ear dose (equivalent to treating a 10 kg child or a 50 kg adult with 0.27 mg or 0.36 mg of ciprofloxacin in each ear twice daily, respectively) did not reveal impairment of fertility or teratogenic effects.
In rabbits, oral ciprofloxacin at 30 and 100 mg/kg did not cause malformations, although significant maternal toxicity was noted. Intravenous administration of doses up to 20 mg/kg did not produce toxic effects on embryos, embryotoxicity, or teratogenicity.
Pharmacokinetics
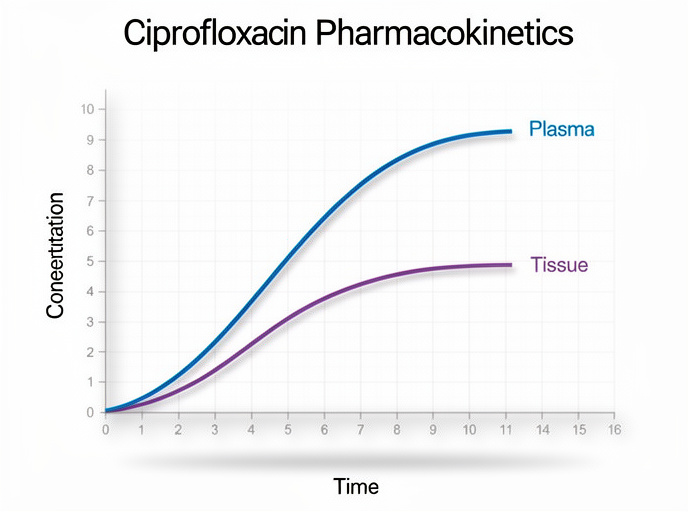
Following topical ocular application in humans, ciprofloxacin is well absorbed into the eye. Concentrations measured in the tear film, cornea, and anterior chamber reach levels ten to several hundred times higher than the MIC90 for sensitive ocular pathogens.
Systemic absorption after topical ocular use is low. After seven days of treatment, plasma concentrations ranged from below the detection limit (< 1.25 ng/mL) up to 4.7 ng/mL. The mean maximum plasma concentration achieved with topical ocular therapy is approximately 450 times lower than that observed after a single 250 mg oral dose.
In children with ear drainage via tympanostomy tube or perforated eardrum, topical otic application resulted in undetectable plasma ciprofloxacin levels (detection limit 5 ng/mL). In animal models (chinchillas), ciprofloxacin appeared in plasma and middle ear fluid after intramuscular injection and penetrated the inner ear following topical administration to the middle ear.
The systemic pharmacokinetics of ciprofloxacin are well characterized: the drug distributes extensively into body tissues, with tissue concentrations generally exceeding plasma levels. The steady-state volume of distribution is approximately 1.7–2.71 L/kg, with serum protein binding of 16–43%. The serum half-life is about 3–5 hours.
After single oral doses of 250–750 mg in adults with normal renal function, 15–50% of the dose is excreted unchanged in urine and 10–15% as metabolites within 24 hours. Both ciprofloxacin and its four major metabolites are eliminated via urine and feces. Renal clearance typically ranges from 300–479 mL/min, and approximately 20–40% of a dose is excreted in the feces as unchanged drug and metabolites within five days.
Clinical indications
Ciprofloxacin eye and ear drops are indicated for the treatment of the following conditions caused by ciprofloxacin-susceptible bacteria:
- Ophthalmic indications: corneal ulcers and superficial infections of the eye and adnexa, including conjunctivitis and blepharoconjunctivitis.
- Otic indications: acute otitis externa and acute otitis media with tympanostomy tube drainage.
Contraindications and precautions
Ciprofloxacin is contraindicated in patients with known hypersensitivity to ciprofloxacin, other quinolone antibiotics, or any component of the formulation.
Because systemic exposure after topical application is low, drug–drug interactions are unlikely. However, as with all antibacterial agents, prolonged use may promote overgrowth of non-susceptible bacteria or fungi. If superinfection occurs, appropriate alternative therapy should be initiated.
Serious and sometimes fatal anaphylactic hypersensitivity reactions have been reported with systemic quinolones, sometimes after the first dose. Reactions may include cardiovascular collapse, loss of consciousness, tinnitus, facial or throat swelling, dyspnea, urticaria, and pruritus. If an acute hypersensitivity reaction occurs with ciprofloxacin drops, treatment should be discontinued immediately and emergency care initiated if clinically indicated (e.g., oxygen therapy and airway management).
Tendinitis and tendon rupture have been associated with systemic fluoroquinolones, particularly in elderly patients and those receiving corticosteroids. Although systemic levels from topical eye/ear therapy are very low, treatment with CILOXAN eye/ear drops should be stopped at the first sign of tendon inflammation.
Eye drops specific precautions: Clinical experience in children under 1 year of age, especially neonates, is limited. Use in newborns with gonococcal or chlamydial ophthalmia neonatorum is not recommended, as it has not been adequately studied. In addition, the risk of drug entering the nasopharynx during instillation should be considered, as this may contribute to bacterial resistance.
CILOXAN eye drops contain benzalkonium chloride, which can irritate and discolor soft contact lenses. Wearing contact lenses during active eye infection and treatment is not recommended; patients should be advised to remove contact lenses before instillation and to avoid using them until therapy is completed and symptoms resolve.
Ear drops specific precautions: The efficacy and safety of ciprofloxacin ear drops in children under 1 year of age have not been fully evaluated. Frequent medical monitoring is recommended to assess treatment response and determine the need for additional therapeutic measures.
Pregnancy, breastfeeding, and driving
There are insufficient data regarding the use of CILOXAN in pregnant women. Animal studies do not indicate direct harmful effects with respect to reproductive toxicity at clinically relevant exposure levels, but, as a precaution, it is generally advisable to avoid using ciprofloxacin eye or ear drops during pregnancy unless the expected benefit clearly outweighs potential risks.
Ciprofloxacin is excreted in breast milk after oral administration. It is unknown whether measurable amounts appear in breast milk after topical ocular or otic use. Therefore, CILOXAN should be used with caution in breastfeeding women, ideally under medical guidance.
This medication has little or no effect on the ability to drive or operate machinery. However, transient blurred vision or other visual disturbances may occur immediately after instillation of eye drops. Patients should wait until vision has cleared before driving or using machines. There are no specific data on the effect of ear drops alone on driving or operating machinery.
Dosage and administration
Ophthalmic use in adults and adolescents (including the elderly): For superficial bacterial infections of the eye and adnexa, the usual dose is 1–2 drops instilled into the conjunctival sac of the affected eye(s) four times daily.
For severe infections, the dosage may be increased to 1–2 drops every two hours during waking hours for the first two days, then reduced according to clinical response. Typical treatment duration ranges from 7 to 14 days.
If other topical ophthalmic medications are used at the same time, an interval of at least 5–15 minutes between applications is recommended to avoid washout and to ensure optimal absorption.
Directions for use: To avoid contamination of the dropper tip and solution, patients should take care not to touch the eyelids, surrounding structures, or any other surfaces with the tip of the bottle. The bottle should be tightly closed after each use.
Overdose
Because ciprofloxacin eye and ear drops are intended for topical external use, toxic effects due to overdose in ophthalmology or otology are not expected when used according to recommendations. Accidental ingestion of the contents of a single vial is unlikely to lead to clinically significant systemic toxicity in individuals with normal renal function.
Adverse reactions
Reported undesirable effects after use of CILOXAN include:
- Local eye or periocular reactions such as stye, eye discomfort, ocular hyperemia, corneal deposits, photophobia, eye itching, increased lacrimation, and eye discharge.
- Systemic or upper respiratory symptoms such as rhinitis and dysgeusia (altered taste).
- Neurological symptoms such as headache and dizziness.
If severe or persistent adverse reactions occur, use of ciprofloxacin should be reassessed and medical advice sought promptly.
How to order ciprofloxacin (Ciloxan) online
You are currently reading an information page about Ciloxan and other ciprofloxacin eye and ear preparations. If you are interested in this medicine and considering an online purchase, it is essential to combine convenience with safe medical practice.
Before you order ciprofloxacin online, review the indications, contraindications, and potential adverse reactions described above and, whenever possible, consult a healthcare professional to confirm that ciprofloxacin is appropriate for your condition. Many reputable online pharmacies provide access to licensed pharmacists or physicians who can answer questions about dosing, interactions, and safe use.
Our partner online store offers CILOXAN and related ciprofloxacin products with mail delivery and discreet packaging. In many jurisdictions, topical ciprofloxacin may be available without a traditional paper prescription, but you should always follow local legal requirements and medical guidance. To explore current prices, available forms, and shipping options, use the Online Store button at the top of this page.
By combining accurate medical information with responsible purchasing behavior, you can use ciprofloxacin eye and ear drops effectively and safely for the treatment of bacterial infections.








